Do you find the topic of acids and bases a confusing one? You’re not alone! This chemistry topic is known to be a tough one, with many tricky equations and things to remember. It is also a very common topic that comes up in many questions in the exams – so it pays to know it well!
If you need help going through the entire topic of acids and bases, our secondary Chemistry tutors will be happy to help! If not, read on for a quick refresher of the most common areas of confusion students have about the topic of acids and bases! (Looking for something else? Maybe our guide on trigonometry can help you!)
Acid concentration vs acid strength
A high concentration of acid means it is a strong acid – true or false?
False! There are two terms to note here: Acid concentration and acid strength.
When we talk about strong and weak acids in terms of acid strength, we are referring to how well the acid ionises. A strong acid like HCl ionizes completely, resulting in a faster rate of reaction, while a weak acid like HCOOH ionizes only partially and reacts at a slower rate.
On the other hand, acid concentration is the amount of acid per unit volume of water. Concentration can also affect the rate of reaction, because a higher concentration means there are more ions to react with the substance.
The strength of an acid is independent of the concentration of it. For example, you can have a high concentration of a weak acid, and the acid reaction will still only occur partially due to the partial ionization of the acid.
Special cases of acid reactions
Acid reactions – easy? We all know there are a few important ones to remember.
But keep in mind a few exceptions to these!
1. Not all metals react with acid
Some metals are unreactive, and thus they do not react with acids. Examples of these are copper and silver.
Some metals will appear not to react with acid, due to the formation of an insoluble compound that forms over the surface of the metal. An example of this is lead. When a piece of lead is put into dilute HCl, insoluble lead (II) chloride will form, covering the surface of the metal and preventing further reaction with the acid.
2. Weak acids do not react completely
You may recall from above that weak acids only react partially. In truth, the reaction is usually in equilibrium, with the reverse reaction also occurring simultaneously.
As a result, the equations for weak acids should have a bidirectional arrow, showing that the chemical reaction is only partial and occurs to an equilibrium.
Base vs alkali
We hear about acids, bases, and alkalis – but what are bases and alkalis, really? To some extent, they sound very similar (they are the ‘opposites’ to acids), but did you know there’s actually a difference?
A base is a metal oxide or hydroxide that reacts with acid to form water and a salt only. In other words, a base is:
- Metal oxide or hydroxide
- Can be soluble or insoluble in water
- Reacts with acid to form water and salt
An alkali is a base that dissolves in water to form hydroxide ions. In other words, alkalis are:
- Water soluble (forms hydroxide ions)
- Metal hydroxides
- Reacts with acid to form water and salt
So, an alkali is a type of base, but not all bases are alkalis!
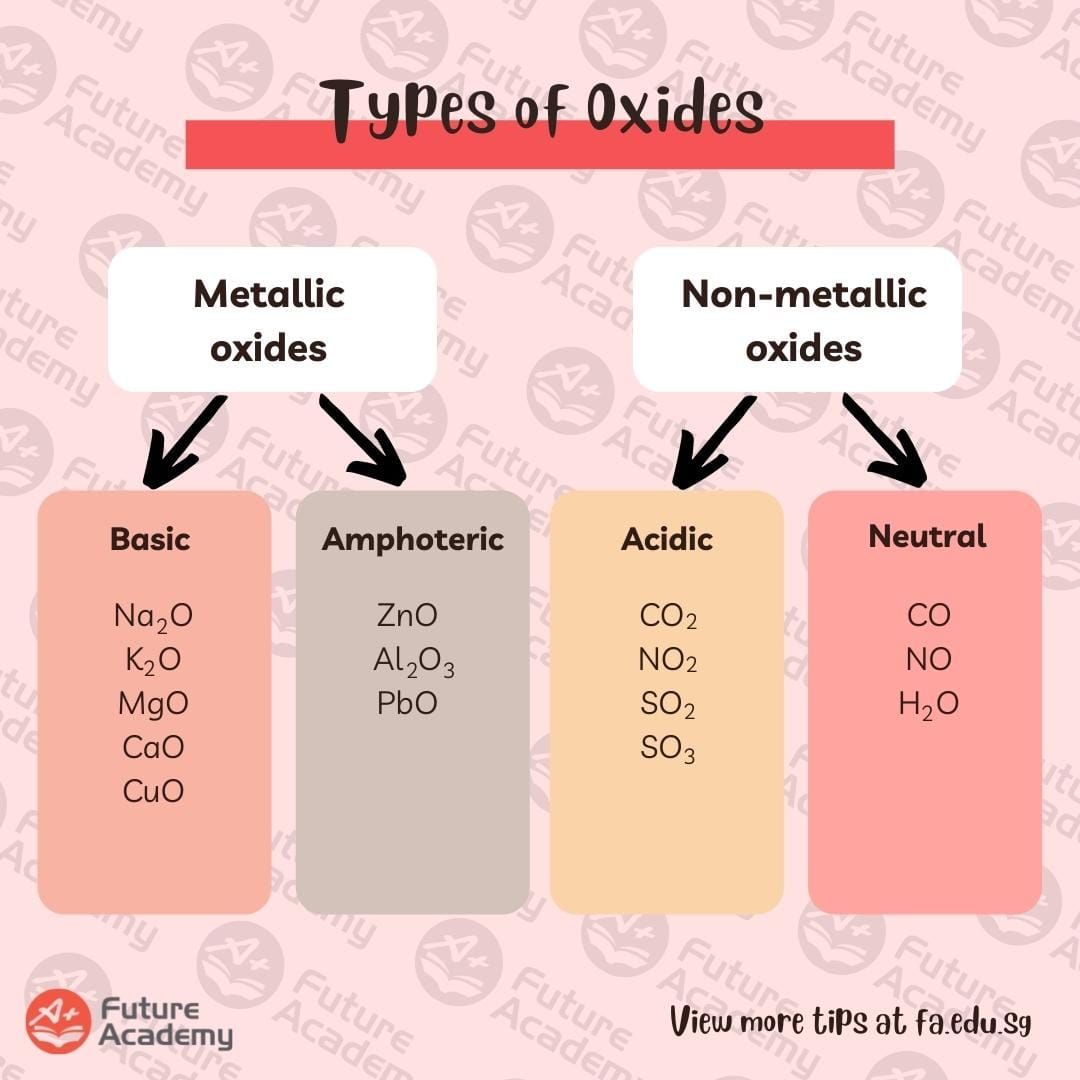
Types of oxides
There are 4 types of oxides, depending on how they behave when reacting with other acids, bases, or when dissolved in water.
One way to recognise the types of oxides is by whether they are metallic or non-metallic.
Metallic oxides are either basic or amphoteric.
Non-metallic oxides are acidic or neutral oxides.
However, what happens if you know a metallic oxide can be basic or amphoteric? How will you know which type it is?
Unfortunately, there isn’t an easy pattern to remember this, you just have to memorise them!
A very, very general pattern we can observe is that the type of oxide from left to right of the periodic table goes from basic oxides to amphoteric oxides.
For non-metallic oxides, observe how the neutral oxides tend to be monoxides.
Conclusion
The difficult thing about chemistry and a topic like acids and bases – there are a lot of rules to remember, and a lot of exceptions to the rules! It often requires intentional memorisation to get everything in your head.
We also believe that getting lots of practice and making this process of learning fun and enjoyable helps immensely! Thankfully, our IP chemistry tutors are known to make classes engaging and beneficial to students.
So, if you or someone you know is in need of chemistry help, look no further! Our expert tutors will be happy to guide you – simply contact us to know more!